Background
Microbial colonization in infants gut influences human physiology, including the maturation of the immune system, nutrient absorption and metabolism, and the protection against pathogen colonization (Buffie and Pamer, 2013). Several factors including the mode of delivery (Dominguez-Bello et al., 2010, 2016), gestational age at birth (La Rosa et al., 2014), maternal and infant antibiotic usage (Lemas et al., 2016) and feeding method (formula or breastfeeding) (Backhed et al., 2015) are very important to the early development of the infant microbiome. Wider environmental exposure (H. Shin et al., 2015) and early intimate relations, particularly with the mother, also play a key role in the early microbial development of an infant. Microbiomes are responsible for different diseases of infants like asthma, diabetes, obesity etc (Nagpal et al. 2018). While the importance of the host-microbiome interplay is not in question, the mechanisms by which an infant acquires these microbes, and from what source, remain largely unexplored. Just a few months ago, a study demonstrated that the maternal microbiome is an important source in the early development of microbial species and strains in the infant gut (Korpela et al., 2018). Yet there has been no comprehensive assessment of the multiple potential maternal sources (like skin, breast milk, fecal, vaginal, oral ect.) of microbial transmission, and how ultimately they contribute to the development of the infant microbiome within hours of birth and over the first few months of life.
Central Question
Ferretti P. and associates (2018) in a recent study focus on how does mother to infant microbial transmission from different maternal body sites help shape the infant gut microbiome? They observed a very high microbial diversity and strain heterogeneity in the pioneering infant gut microbiome even in the first day of life, and that diversity dramatically decreases within the first week before recovering and gradually increasing over the next 4 months. In addition, they identified microbial strains present in the infants that are transmitted from their mothers, and these strains are more likely to adapt and persist in the infant gut than non-maternally acquired strains.
Evidences
Early Microbial Diversity In The Infant Gut Is Shaped By Microbial Transmission From Maternal Body Sites
The authors observed a high diversity in the infant fecal microbiomes within 24 hours of birth. This diversity decreased over the first week postpartum before recovering over time. The initial high diversity is due to the rapid transmission of microbes from maternal as well as other environmental sources after birth, and this information is supported by a previous report using gene sequencing (Wampach et al., 2017). To add support to vertical mother-to-infant transmission events, the authors implemented a novel combination of metagenomic strain-profiling tools, expanding on methods validated previously (Asnicar et al., 2017). With this approach, they detected a total of 52 strains shared within mother and infant pairs (out of 317 cases of shared mother-infant species with typable strains), i.e., a 16.4% strain transmission rate. By comparison, they found 46 strains in infants that were in common with unrelated mothers out of a total of 6,319 mother-infant inter-pair shared species. Some of these microbes are present for a limited period of time in some infants as they are not suited to colonize the infant gut. Others are true early colonizers. Among the species that were lost in the infants between day 1 and the subsequent time periods, 80% were shared with at least one body site of the respective mother.
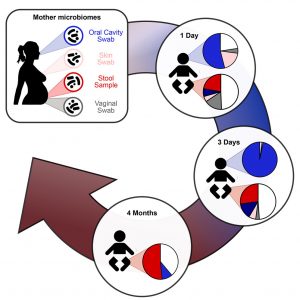
Enriched Maternal Microbial Species In Infants Body
The infant gut microbiomes displayed a large proportion of species in common with their mothers for all mother-infant pairs. In the first day of birth, about 50% of the microbial population in the infant gut belonged to species also present in at least one of the sampled maternal body sites. And the study found that this percentage remains relatively stable over time (around 50% from day 1 to 4 months). Considering the maternal sources of transmission, the study found the gut microbiome as the largest donor of the infant-acquired strains. The number of strains in common between mother and infant gradually decreases over time, with 23 at 1 day and 28 at 3 days, compared with 10 at 1 week, 10 at 1 month, and 6 at 4 months. This supports the hypothesis of selection for niche-specific bacteria from the pool of maternal strains seeding the infant gut.
Future Research
This study quite remarkably find out the microbial diversity of an infant and the way of mother to infant microbial transmission. And these study findings may help a lot in the upcoming research on this topic. Past research showed that delivery mode and feeding can shape the bacterial community in the infant gut (Azad et al. 2013). Knowing this, a future study can look at how this microbial transmission differs depending upon the mode of delivery (Caesarean delivery/ Vaginal delivery) of infant. In addition, I would like to research on the other possible potential microbial sources of transmission, such as father, siblings and environments (hospital and house surfaces) and their effect on development of the infant gut microbiome.
Further Reading
The following article have more details about mother to child microbial transmission events. In addition it analyzed the incidence of family specific antibiotic resistance genes during maternal bacterial transmission. So if you have interest on those topics you should have a look on it.
Yassour, M., Jason, E., Hogstrom, L. et al. Strain-level analysis of mother-to-child bacterial transmission during the first few months of life. Cell Host Microbe 24, 146—154 (2018). doi: 10.1016/j.chom.2018.06.007
The following article is based on findings from a pilot study on how the infant gut is colonized from maternal sources and ultimately develops an adult like composition. If you have interest on early maternal microbial transmission then you should read the following article:
Asnicar, F. et al. Studying Vertical Microbiome Transmission from Mothers to Infants by Strain-Level Metagenomic Profiling (2017). doi: 10.1128/MSYSTEMS.00164-16
References
- Asnicar, F., Manara, S., Zolfo, M., Truong, D.T., Scholz, M., Armanini, F., Ferretti, P., Gorfer, V., Pedrotti, A., Tett, A., et al. (2017). Studying vertical microbiome transmission from mothers to infants by strain-level metagenomic profiling. mSystems 2. doi: 10.1128/mSystems.00164-16
- Azad, M. B., Konya, T., Maughan, H., Guttman, D. S., Field, C. J., Chari, R. S., Kozyrskyj, A. L. (2013). Gut microbiota of healthy Canadian infants: profiles by mode of delivery and infant diet at 4 months. CMAJ’¯: Canadian Medical Association Journal, 185(5), 385—394. doi: 10.1503/cmaj.121189
- Backhed, F., Roswall, J., Peng, Y., Feng, Q., Jia, H., Kovatcheva-Datchary, P., Li, Y., Xia, Y., Xie, H., Zhong, H., et al. (2015). Dynamics and stabilization of the human gut microbiome during the first year of life. Cell Host Microbe 17, 690—703. doi: 10.1016/j.chom.2015.04.004
- Buffie, C.G., and Pamer, E.G. (2013). Microbiota-mediated colonization resistance against intestinal pathogens. Nat. Rev. Immunol. 13, 790—801. doi: 10.1038/nri3535
- Dominguez-Bello, M.G., Costello, E.K., Contreras, M., Magris, M., Hidalgo, G., Fierer, N., and Knight, R. (2010). Delivery mode shapes the acquisition and structure of the initial microbiota across multiple body habitats in newborns. Proc. Natl. Acad. Sci. USA 107, 11971—11975. doi: 10.1073/pnas.1002601107
- Dominguez-Bello, M.G., De Jesus-Laboy, K.M., Shen, N., Cox, L.M., Amir, A., Gonzalez, A., Bokulich, N.A., Song, S.J., and Hoashi, M. (2016). Partial restoration of the microbiota of cesarean-born infants via vaginal microbial transfer. Nat. Med. 22, 250—253. doi: 10.1038/nri3535
- Ferretti P, Pasolli E, Tett A, et al. Mother-to-infant microbial transmission from different body sites shapes the developing infant gut microbiome. Cell Host Microbe. 2018; 24(1):133-45. doi: 10.1016/j.chom.2018.06.005.
- H. Shin, Z. Pei, K.A. Martinez 2nd, J.I. Rivera-Vinas, K. Mendez, H. Cavallin, M.G. Dominguez-Bello (2015). The first microbial environment of infants born by C-section: the operating room microbes. Nat. Med., 3, p. 59. doi: 10.1186/s40168-015-0126-1
- Korpela, K., Costea, P., Coelho, L.P., Kandels-Lewis, S., Willemsen, G., Boomsma, D.I., Segata, N., and Bork, P. (2018). Selective maternal seeding and environment shape the human gut microbiome. Genome Res. 28, 561—568. doi: 10.1101/gr.233940.117
- La Rosa, P.S., Warner, B.B., Zhou, Y., Weinstock, G.M., Sodergren, E., HallMoore, C.M., Stevens, H.J., Bennett, W.E., Jr., Shaikh, N., Linneman, L.A., et al. (2014). Patterned progression of bacterial populations in the premature infant gut. Proc. Natl. Acad. Sci. USA 111, 12522—12527. doi: 10.1073/pnas.1409497111
- Lemas, D.J., Yee, S., Cacho, N., Miller, D., Cardel, M., Gurka, M., Janicke, D., and Shenkman, E. (2016). Exploring the contribution of maternal antibiotics and breastfeeding to development of the infant microbiome and pediatric obesity. Semin. Fetal Neonatal Med. 21, 406—409. doi: 10.1016/j.siny.2016.04.013
- Nagpal R, Yamashiro Y: Gut Microbiota Composition in Healthy Japanese Infants and Young Adults Born by C-Section. Ann Nutr Metab 2018;73(suppl 3):4-11. doi: 10.1159/000490841
- Wampach, L., Heintz-Buschart, A., Hogan, A., Muller, E.E.L., Narayanasamy, S., Laczny, C.C., Hugerth, L.W., Bindl, L., Bottu, J., Andersson, A.F., et al. (2017). Colonization and Succession within the human gut microbiome by archaea, bacteria, and microeukaryotes during the first year of life. Front. Microbiol. 8, 738. doi: 10.3389/fmicb.2017.00738
