INTRO:
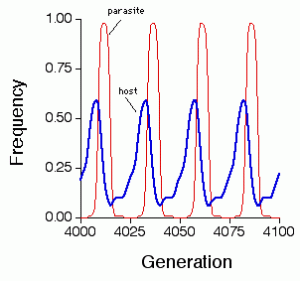
Viruses and their hosts influence each other’s evolution. As selection acts on viruses to increase their ability to infect hosts, selection is acting on hosts to decrease their susceptibility to viruses, and the next generation of viruses is less effective at infecting hosts. But the viruses continue to evolve, as do the hosts–they coevolve and often demonstrate this pattern:
This is called the Red Queen Effect, after the Red Queen from Through the Looking Glass by Lewis Carroll.

Innate immunity is one of the adaptations hosts have evolved to combat viruses. Host cells produce signaling proteins called interferons to indicate the presence of viruses and call for antiviral help. Innate immunity of hosts is an important pressure shaping the evolution of viruses. If viruses are able to overcome the innate immunity of hosts and infect them, they are able to survive and reproduce. Host species are another pressure shaping virus adaptation. Viruses that can infect multiple hosts will have more opportunity to reproduce, but different hosts have different defenses that require different things for the viruses to overcome. The number of hosts a virus is able to infect is called it’s host breadth.
Host species as a pressure shaping viral evolution is relevant to human health. HIV has been traces back to simian immunodeficiency virus (SIV) in chimpanzees. An infected chimp had contact with a human and this particular strain of the virus was able to infect human cells. Wasik et. al. explore the these two pressures on the adaptations of viruses, host species and innate immunity.
BIOLOGICAL SYSTEM:
To explore virus-host coevolution and innate immunity and host species, a virus was evolved on several cell types in the lab.
Virus-Vesicular Stomatitis Virus (VSV) lineages were evolved on the following host cell types:
- Deficient host: HeLa (Human carcinoma cells with innate immune-deficient function)
- Competent host: MDCK (Canine kidney cells with innate immunity intact)
- Alternating Host (HeLa and MDCK)
The viruses grown on the deficient host cells wouldn’t have to respond to the pressures of innate immunity, and so were predicted to be less fit in an environment where the host cells have innate immunity. The cells grown up on the competent MDCK cells would be pressured to overcome innate immunity, but were predicted to be able to infect immune-deficient cells as well.
EXPERIMENT:
To test how innate immunity and host species affected viral populations, the viruses were introduced to the four environments:
- VSV evolved in HeLa was introduced to LNCaP (Human prostate cancer cells, innate immune-deficient) and PC-3 (human prostate cancer cells, innate immune-competent).
- Virus evolved in MDCK was introduced to LNCaP and PC-3.
- Virus evolved in alternating HeLa and MDCK was introduced to LNCaP and PC-3.
- HeLa-evolved viruses on innate immune-deficient cells of nonhuman primate origin (VERO African green monkey kidney cells).
How well the viruses were able to infect each cell type was measured as fitness.
KEY RESULTS:
The results for the first three experiments are shown below.
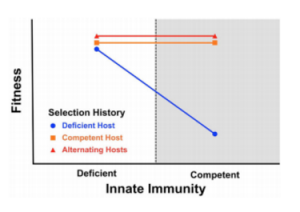
The points in the white area represent the fitness of the virus on immune deficient host cells, and the points in the shaded area represent the fitness of the virus on immune competent host cells. The strains of the virus that evolved on immune competent hosts or alternating competent and deficient hosts (represented by orange squares and red triangles respectively) do not vary significantly in fitness regardless of their host’s immune competence. However, the virus that evolved on immune deficient host cells is significantly less fit on immune competent hosts.
The figure below shows the results from the experiment testing the effects of host species on viral fitness.
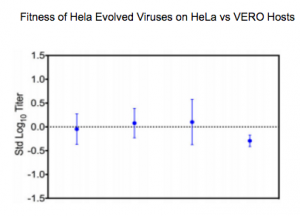
The points represent the fitness of viruses evolved on immune deficient human host cells on those same host cells compared to the fitness on immune deficient monkey host cells. A value of zero indicates no difference in fitness; these results indicate that host species had very little effect on viral fitness.
Fitness of the viruses was primarily defined by the host’s innate immune response. In other words, it mattered more whether the host had innate immunity than what species it was. “Innate immunity is more prominent than host species in determining viral fitness at the host cell level.’
FURTHER DIRECTIONS:
Further directions in this realm of research include:
- Understanding the key genetic differences (what new alleles arise and what their functions are) in viruses evolved on immunocompetent vs immunodeficient and alternating host cells.
- Exploring how host-immune competence and other host factors affect virus genetic diversity.
- Exploring how genetic diversity in viruses affects viral virulence and the ability to opportunistically transmit to new hosts.
Some of these directions are explored through modeling by Nuismer and Dybdahl. They modeled host’s innate immunity as a two locus system, with locus A determining whether or not the host recognized the virus, and locus B determining to what how much of an attack they launched on the virus. They explored how much diversity within populations was maintained for locus A compared to locus B, interactions between host genotype and virus genotype with the two locus model, and how each locus affected local adaptation of viruses. Translating some of their models into wet-lab experiments could be a direction for further research.
REFERENCES:
https://www.theaidsinstitute.org/education/aids-101/where-did-hiv-come-0